Redox flow batteries are one of the most promising technologies for large-scale stationary storage applications due to their low capital cost, low flammability, and long lifetime of over 20 years. However, since the price of vanadium, the most widely used active material for redox flow batteries, has been rising in recent years, scientists have been actively searching for redox materials to replace it.
Now a research team from the Korea Advanced Institute of Science and Technology (KAIST) and Pohang University of Science and Technology (POSTECH) in South Korea has developed a highly soluble and stable active molecule – naphthalene diimide (NDI). It was used in place of vanadates in aqueous flow batteries.
Although NDI molecules are almost insoluble in water and were therefore little explored, the South Korean research team managed to tether four ammonium functionalities and achieved a solubility as high as 1.5M in water. By regulating the π–π interactions of these organic molecules, the researchers have avoided any severe side reactions and reduced cyclability which could have been caused by radical formation during the electron transfer process.
“We have demonstrated the principles of molecular design by modifying an existing organic active molecule with low solubility and utilizing it as an active molecule for redox flow batteries,” Professor Hye Ryung Byon said. “We have also shown that during a redox reaction, we can use molecular interactions to suppress the chemical reactivity of radically formed molecules.”
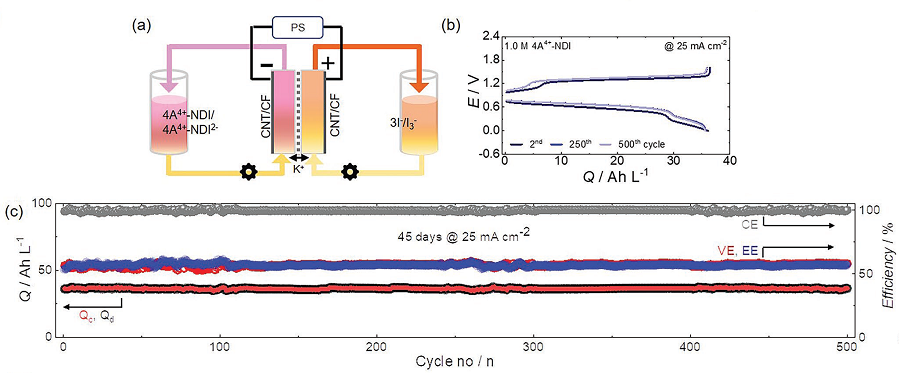
Furthermore, the researchers showed that when a 1M solution of NDI was used in neutral redox flow batteries for 500 cycles, 98% of its capacity was maintained. This means 0.004% capacity decay per cycle, and only 2% of its capacity would be lost if the battery were to be operated for 45 days.
Popular content
The researchers also showed that the developed NDI molecule can hold two electrons per molecule, meaning that 2M of electrons could be stored in every 1M of NDI solution used.
For reference, vanadium used in vanadium redox flow batteries, which require a highly concentrated sulfuric acid solution, has a solubility of about 1.6M and can only hold one electron per molecule, meaning it can store a total of 1.6M of electrons. Therefore, the newly developed NDI active molecule shows a higher storage capacity compared to existing vanadium devices.
“Should this be used later for aqueous redox flow batteries, along with its high energy density and high solubility, it would also have the advantage of being available for use in neutral pH electrolytes,” Ryung Byon said. “Vanadium redox flow batteries currently use acidic solutions, which cause corrosion, and we expect our molecule to solve this issue. Since existing lithium ion-based ESS are flammable, we must develop safer and cheaper next-generation ESS, and our research has shown great promise in addressing this.”
The researchers discussed their findings in the paper “Controlling π-π interactions of highly soluble naphthalene diimide derivatives for neutral pH aqueous redox flow batteries,” which was recently published in Advanced Materials.
This content is protected by copyright and may not be reused. If you want to cooperate with us and would like to reuse some of our content, please contact: editors@pv-magazine.com.


By submitting this form you agree to pv magazine using your data for the purposes of publishing your comment.
Your personal data will only be disclosed or otherwise transmitted to third parties for the purposes of spam filtering or if this is necessary for technical maintenance of the website. Any other transfer to third parties will not take place unless this is justified on the basis of applicable data protection regulations or if pv magazine is legally obliged to do so.
You may revoke this consent at any time with effect for the future, in which case your personal data will be deleted immediately. Otherwise, your data will be deleted if pv magazine has processed your request or the purpose of data storage is fulfilled.
Further information on data privacy can be found in our Data Protection Policy.