Many leading research institutes the world over are looking into artificial photosynthesis processes, which can use energy from sunlight to drive a chemical reaction that splits water to generate hydrogen.
Such devices have not yet achieved the type of efficiency that would make them commercially viable, and often require an additional power source to drive the reaction, meanwhile powering an electrolyzer to produce hydrogen from solar generated electricity is a concept that is rapidly gaining commercial ground.
Now, however, researchers at the Lawrence Berkeley Lab (LBL) in California, working alongside the Joint Center for Artificial Photosynthesis have successfully demonstrated a device which combines a hydrogen producing photoelectrochemical cell with a silicon PV cell.
The device developed by LBL is described in the paper Hybrid photoelectrochemical and photovoltaic cells for simultaneous production of chemical fuels and electrical power, published in the journal Nature Materials. The researchers calculate that such a device could achieve 6.8% hydrogen efficiency, and 13.4%, putting a combined 20.2% of sunshine hitting the cell to use.
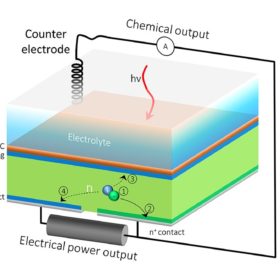
Image: Berkeley Lab/JCAP
The problem with such devices in the past, according to LBL, is that the lower performance of the other materials in the stack limit the current that can be generated in the silicon cell. “It's like always running a car in first gear,” explains Gideon Segev, the study’s lead author. “This is energy that you could harvest, but because silicon isn't acting at its maximum power point, most of the excited electrons in the silicon have nowhere to go, so they lose their energy before they are utilized to do useful work.”
Popular content
The researchers got around this problem by simply adding an additional contact to the rear of the silicon cell, which could split the current in two – with one part contributing to the hydrogen production, and the other free to harvest more of the power generated in the silicon cell.
“The concept that photogenerated charge carriers can be controllably directed to produce electricity and chemical fuel provides an opportunity to significantly increase the energy return on energy invested in solar fuels systems,” reads the paper’s abstract, “and can be adapted to a variety of architectures assembled from different materials.” Segev says that the researchers plan to continue their collaboration and look into further applications for the hybrid device.
This article was updated on 01.11.2018, to correct a statement that prototype devices had actually achieved the quoted efficiencies, which are in fact only the result of calculations.
This content is protected by copyright and may not be reused. If you want to cooperate with us and would like to reuse some of our content, please contact: editors@pv-magazine.com.



By submitting this form you agree to pv magazine using your data for the purposes of publishing your comment.
Your personal data will only be disclosed or otherwise transmitted to third parties for the purposes of spam filtering or if this is necessary for technical maintenance of the website. Any other transfer to third parties will not take place unless this is justified on the basis of applicable data protection regulations or if pv magazine is legally obliged to do so.
You may revoke this consent at any time with effect for the future, in which case your personal data will be deleted immediately. Otherwise, your data will be deleted if pv magazine has processed your request or the purpose of data storage is fulfilled.
Further information on data privacy can be found in our Data Protection Policy.