Lithium-ion batteries have key roles to play across several areas of the energy transition. And while they are already doing this, and progress has been rapid, the technology still has a host of challenges to overcome. Not least is increasing the lifetime of batteries to match expectations for vehicles, energy infrastructure, and other devices they will be powering.
Batteries with lithium-ion chemistry are affected by several degradation mechanisms that can cause them to lose capacity over time. In many cases, these involve oxygen, with battery components becoming oxidized and with reduced capacity for the desired chemical reactions that amount to energy storage. Various interface-engineering and other strategies have been employed to reduce oxidation during battery cycling but, for the most part, they have only been able to slow the inevitable.
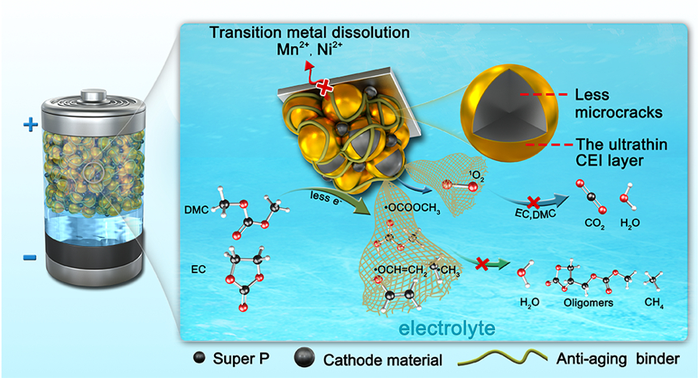
A group of scientists led by the Qingdao Institute of Bioenergy and Bioprocess Technology (QIBEBT), in China, noted oxidation reactions happen in a lot of places other than inside batteries and, given the abundance and reactivity of oxygen, such reactions are one of the most common types of chemical degradation on the planet. In many cases, organisms have naturally developed processes to limit oxidation and related degradation and aging processes. “So, we thought: ‘why not just try to replicate what nature already does, and put it inside a battery instead?'” said QIBEBT researcher Cui Guanglei.
Inspired by nature
The group developed an additive known as a photostabilizer, which they showed could ‘scavenge’ oxygen particles which would otherwise contribute to degradation. With calculations and experiments, they showed such an additive could deliver improved electrochemical performance in batteries and also worked at elevated temperatures. The research and results are described in full in the paper Bioinspired Antiaging Binder Additive Addressing the Challenge of Chemical Degradation of Electrolyte at Cathode/Electrolyte Interphase, published in the Journal of the American Chemical Society.
“This bioinspired strategy to scavenge reactive oxygen and free radicals heralds a new paradigm for manipulating the cathode/electrolyte interphase chemistry of various rechargeable batteries involving layered transition metal oxides-based cathodes,” said Cui.
The group says it now plans to explore commercialization of its approach in high-voltage li-ion batteries based on layered oxide cathodes, to produce “the next generation of energy storage devices beyond traditional lithium-ion technology.”
This content is protected by copyright and may not be reused. If you want to cooperate with us and would like to reuse some of our content, please contact: editors@pv-magazine.com.



Would this be useful for solid-state cells, or is oxidative damage in them not a problem?
Would this be applicable to perovskite PV cells?