A Swiss-Japanese research group has utilized ionic liquids (ILs) with halide anions as additives to improve the performance and stability of a perovskite solar cell. Ionic liquids are considered a valid and greener alternative to organic solvents due to their lower volatility and flammability, as well as to their wide liquid-state window.
Their work built on previous research showing that ILs doping containing iodide anions or cations with fluorous chains did not influence the overall 3D structure of perovskite films, with the stability of the solar cells being increased by the formation of the hydrophobic surface. “As ILs are gaining increased popularity as additives for perovskite solar cells because of their attractive physical properties, such as non-volatility and high thermal and electrochemical stability, the chemical mechanism of the passivation process is an urgent issue that has been long overdue in being tackled,” the scientists explained.
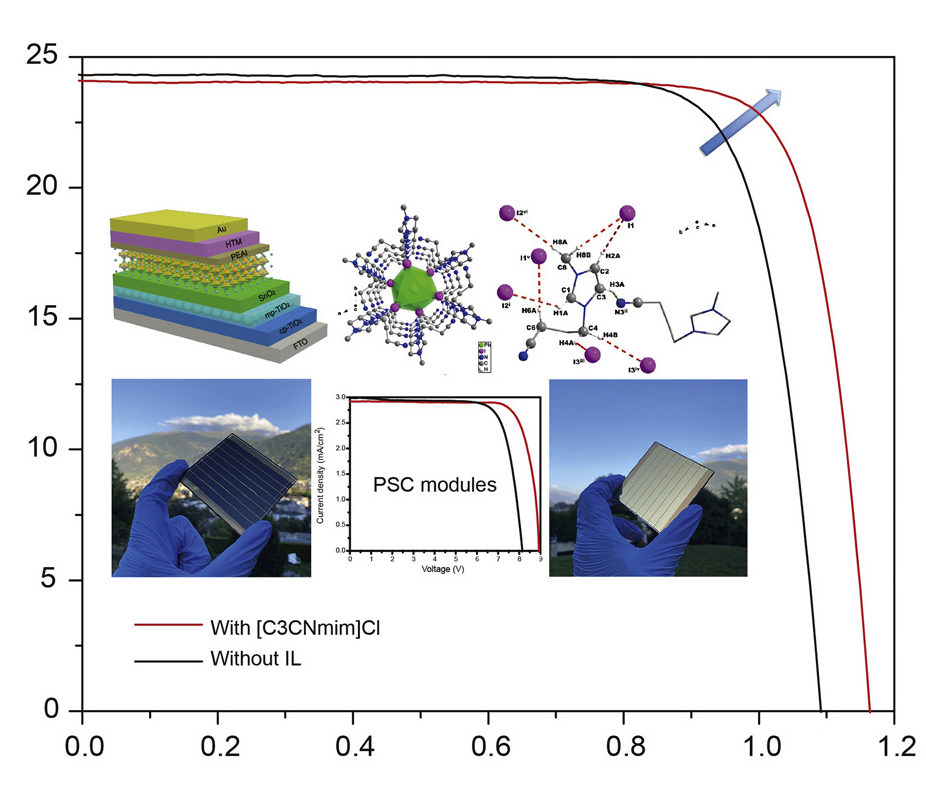
Three different ILs, ([C1CNmim]Cl, [C3CNmim]Cl, and [(C3CN)2im]Cl), were paired with a chloride anion and utilized as additives for a perovskite precursor solution known as (Cs0.08FA0.8MA0.12)Pb(I0.88Br0.12)3. Chloride salts were employed for the first time in the chloride anion to stabilize the perovskite crystal lattice. The three resulting pervoskite films, according to the academics, exhibited improved uniformity and clear grain morphology with bigger crystal size compared to reference films without ILs.
The films were integrated in a solar cell architecture consisting of a a fluorine-doped tin oxide (FTO) substrate, a titanium dioxide (TiO₂) layer, a Tin(IV) oxide (SnO2) layer, the perovskite layer, a hole transporting material made of phenethylammonium iodide (PEAI), and a layer made of gold (Au). “For all PSC devices, the perovskite absorber layer was prepared via a one-step spin-coating method,” the scientists emphasized. “All of the perovskite films were treated with PEAI prior to the deposition of the HTMs, a recent but widely used technique that results in increased performance.”
The cells were tested under standard illumination conditions and the device based on [C3CNim]Cl achieved the highest power conversion efficiency of 22.86%, an open-circuit voltage of 1.16 V, a short-circuit current of 24.03 (mA cm−2), and a fill factor of 0.818. The reference cell without ILs, by comparison, achieved an efficiency of 20.94%, an open-circuit voltage of 1.09 V, a short-circuit current of 24.31 of mA cm−2, and a fill factor of 0.793. The other two cells fabricated with [C1CNim]Cl and [(C3CN)2im]Cl achieved efficiencies of 21.82% and 22.45, respectively.
With the champion cell, the Swiss-Japanese team fabricated a mini module with a size of 7 cm × 6.5 cm that showed an efficiency of 20.9%, which compares to 18.54% in a reference module without the IL-based device. The panel also showed remarkable stability, with around 95% of its initial efficiency being retained after 1,000 hours of operation.
Their findings are presented in the paper “Halide exchange in the passivation of perovskite solar cells with functionalized ionic liquids,” published in Cell Reports Physical Science. The research group is formed by scientists from the University of Tokyo and Toyota Motor Corporation in Japan, and the Swiss Federal Institute of Technology Lausanne (EPFL).
This content is protected by copyright and may not be reused. If you want to cooperate with us and would like to reuse some of our content, please contact: editors@pv-magazine.com.



By submitting this form you agree to pv magazine using your data for the purposes of publishing your comment.
Your personal data will only be disclosed or otherwise transmitted to third parties for the purposes of spam filtering or if this is necessary for technical maintenance of the website. Any other transfer to third parties will not take place unless this is justified on the basis of applicable data protection regulations or if pv magazine is legally obliged to do so.
You may revoke this consent at any time with effect for the future, in which case your personal data will be deleted immediately. Otherwise, your data will be deleted if pv magazine has processed your request or the purpose of data storage is fulfilled.
Further information on data privacy can be found in our Data Protection Policy.