Scientists at the University of California, Riverside, have discovered a method for controlling electron transfer, and guiding electrons in one direction. Electron transfer is one of the most important processes in a solar cell, as well as in batteries, fuel cells, photosynthesis and many other vital processes.
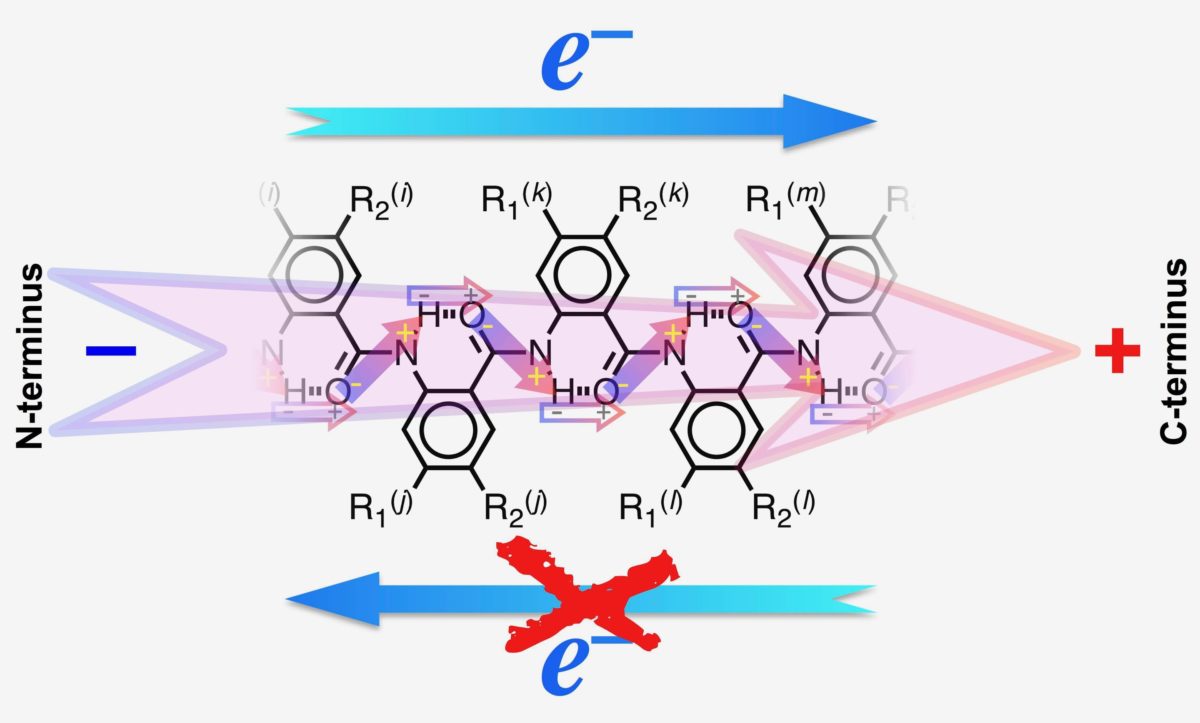
The research relies on electric dipoles, which occur when an atom in a molecule has a composition that is more likely to attract electrons. Dipoles create powerful electric fields at the nano-scale, which can guide electron transfer processes, and suppress undesired ones.
In their paper, ‘Dipole Effects on Electron Transfer are Enormous’ published in the journal Angewandte Chemie International Edition, the researchers describe incorporating an electric dipole into an electron donor molecule, in this case 5-N-amido-anthranilamide electret, a substance with a semi-permanent electric charge and dipole polarization, similar to a magnet.
According to the researchers, these results represent the first time that scientists have used dipoles to encourage electron transfer in one direction, whilst completely suppressing it in the other, which they go on to describe as one of the ‘holy grails of photophysics and energy science.’
“While it appears that we are solving an important physical chemistry and physics problem, the findings from our work can have broad interdisciplinary impacts, and prove important for pertinent fields, such as molecular biology, cell physiology, and energy science and engineering,” said Valentine Vullev, a professor of bioengineering in the Bourns College of Engineering, who led the research team. “A better understanding of electron transfer at the molecular level will improve our understanding of living systems and serve as a foundation for efficient energy technologies.”
This content is protected by copyright and may not be reused. If you want to cooperate with us and would like to reuse some of our content, please contact: editors@pv-magazine.com.



By submitting this form you agree to pv magazine using your data for the purposes of publishing your comment.
Your personal data will only be disclosed or otherwise transmitted to third parties for the purposes of spam filtering or if this is necessary for technical maintenance of the website. Any other transfer to third parties will not take place unless this is justified on the basis of applicable data protection regulations or if pv magazine is legally obliged to do so.
You may revoke this consent at any time with effect for the future, in which case your personal data will be deleted immediately. Otherwise, your data will be deleted if pv magazine has processed your request or the purpose of data storage is fulfilled.
Further information on data privacy can be found in our Data Protection Policy.