Scientists at the Eindhoven University of Technology have come up with a thermodynamic theory that, in their view, explains why perovskite solar cells suffer from thermal stability, which is a crucial issue to overcome in order to take this cell technology out of the laboratories and nearer to commercial production.
In the study Unified theory for light-induced halide segregation in mixed halide perovskites, published in nature communications, the Dutch group explained that the vast majority of perovskites used for PV applications are halide compounds combining organic materials such as methylammonium or formamidinium with a metal, which usually is lead or tin, and halides, like bromide or iodide.
Their analysis was focused on five types of halide compounds combining both bromide and iodide. “This combination works particularly well because it allows for the ‘tuning’ of the bandgap, or the minimum amount of photon energy needed to generate electricity in the material,” they further explained, adding that this solution is ideal when perovskite PV devices are used in tandem solar cells.
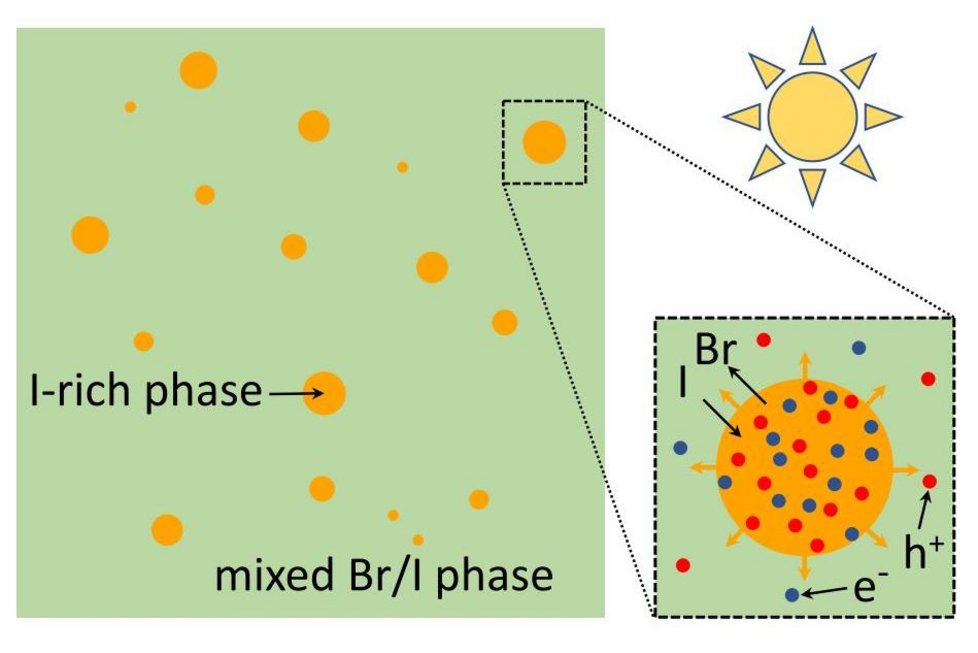
In halide compounds, however, it is frequent that iodide-rich regions are spontaneously formed and bromide is expelled from these regions – a phenomenon which is known as halide segregation. “The subsequent segregation of the compound tends to trap the electricity-producing photocarriers in these low-bandgap areas, severely hampering the efficiency of the cell,” the academics highlighted, noting that the excited photocarriers tend to move to areas where their free energy is lowest.
For each of the five compounds analyzed, the researchers were able to identify stable, metastable, and unstable regions depending on temperature, light conditions, and bromide concentration. “Our unified theory for light-induced halide segregation looks at the total free energy of the perovskite in the photovoltaic cells, both in the dark and when the cell is exposed to sunlight,” stated researcher Peter Bobbert.
The academics claim that this theory may provide technical solutions to build more stable perovskite PV devices. For example, it could help determine how much bromide you can add to the compound without making it unstable. “By not mixing in an excessive amount of bromide, you can avoid segregation while at the same time still achieving a reasonably large bandgap that works for tandem cells,” Bobbert added.
The scientists also found that replacing organic cations with cesium may also have a stabilizing effect. They worked on a cesium-lead compound cell that is claimed to be stable up to 42% bromide concentration and have a maximum bandgap of 1.94 eV, which would be enough to make this cell applicable in tandem PV devices. “The theory can also be readily applied to other semiconductors where the bandgap is tuned by alloying,” they concluded.
This content is protected by copyright and may not be reused. If you want to cooperate with us and would like to reuse some of our content, please contact: editors@pv-magazine.com.



By submitting this form you agree to pv magazine using your data for the purposes of publishing your comment.
Your personal data will only be disclosed or otherwise transmitted to third parties for the purposes of spam filtering or if this is necessary for technical maintenance of the website. Any other transfer to third parties will not take place unless this is justified on the basis of applicable data protection regulations or if pv magazine is legally obliged to do so.
You may revoke this consent at any time with effect for the future, in which case your personal data will be deleted immediately. Otherwise, your data will be deleted if pv magazine has processed your request or the purpose of data storage is fulfilled.
Further information on data privacy can be found in our Data Protection Policy.