Researchers at Harvard claim to have developed a lithium-metal, solid-state battery with improved stability and increased ability to prevent lithium (Li) dendrite penetration.
“Our research shows that the solid-state battery could be fundamentally different from the commercial liquid electrolyte lithium-ion battery,” said researcher Xin Li. “By studying their fundamental thermodynamics, we can unlock superior performance and harness their abundant opportunities.”
The battery can be charged and discharged at least 10,000 times at high current density, the scientists said. Its multilayer design is described as a structure in which a less-stable electrolyte is sandwiched between more stable solid electrolytes that are able to prevent any lithium dendrite growth. This architecture was then combined with a commercial high energy density cathode material.
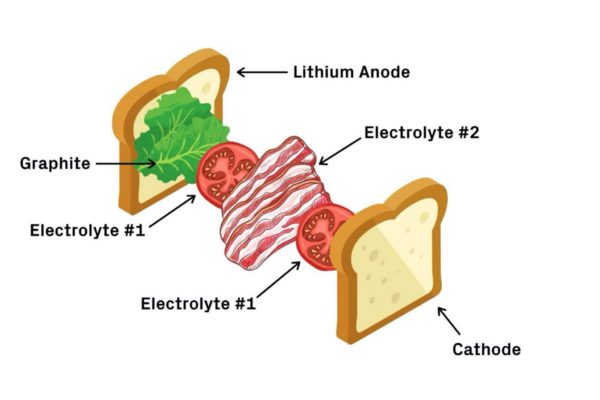
Image: Harward University
“Think of the battery like a BLT sandwich,” the Harvard group explained. “First comes the bread — the lithium metal anode — followed by lettuce — a coating of graphite. Next, a layer of tomatoes — the first electrolyte — and a layer of bacon — the second electrolyte. Finish it off with another layer of tomatoes and the last piece of bread — the cathode.”
The first electrolyte, which the academics designated with the chemical name Li5.5PS4.5Cl1.5, or LPSCI, is more stable with lithium but subject to dendrite penetration. By contrast, the second one, dubbed Li10Ge1P2S12, or LGPS, is more immune to dendrites despite being less stable with lithium. Under this configuration, dendrites are allowed to pass through the graphite and first electrolyte but are stopped right before the second.
“The cycling performance of the lithium metal anode paired with a LiNi0.8Mn0.1Co0.1O2 cathode is very stable, with an 82 per cent capacity retention after 10,000 cycles at a 20C rate and 81.3 per cent capacity retention after 2,000 cycles at a 1.5C rate,” the academics emphasized. “Our design also enables a specific power of 110.6 kilowatts per kilogram and specific energy up to 631.1 watt hours per kilogram at the micrometre-sized cathode material level.”
The battery, according to the academics, also has self-healing properties, as it should be able to backfill holes created by the dendrites. Its creators claim it has the potential to recharge electric vehicles within 10 to 20 minutes.
The device design was described in the paper “A dynamic stability design strategy for lithium metal solid-state batteries”, published in nature.
This content is protected by copyright and may not be reused. If you want to cooperate with us and would like to reuse some of our content, please contact: editors@pv-magazine.com.



when it market the solid state battery?
Reading the Nature article, their solid-state battery cell requires a minimum pressure of 500 atmospheres or 7250 psi. They used up to 2500 atmospheres and 36000 psi in their data. It is a small detail not mentioned in the abstract. This cell is entirely useless for any practical EV application. Any practical cell can’t require more than 7 atmospheres or 100 psi, and even that complicates the design and reduces the energy density quite a bit by requiring pressure plates and screws.