Scientists from Pusan National University in South Korea have looked at how hydrogen could be stored in cryogenic liquid fuel tanks. They found that vaporization and thermodynamic changes represent two crucial factors in this process.
“Despite thermal insulation, the liquefied fuel in a cryotank experiences a degree of vaporization,” they explained, noting that high vaporization, measured as Boil-Off Gas (BOG), can result in excess internal pressure inside the tanks, leading to cracks and fissures.
“This makes understanding and controlling BOG a key factor in cryogenic tank design,” they said in “Experimental and numerical investigation of change in boil-off gas and thermodynamic characteristics according to filling ratio in a C-type cryogenic liquid fuel tank,” which was recently published in Energy.
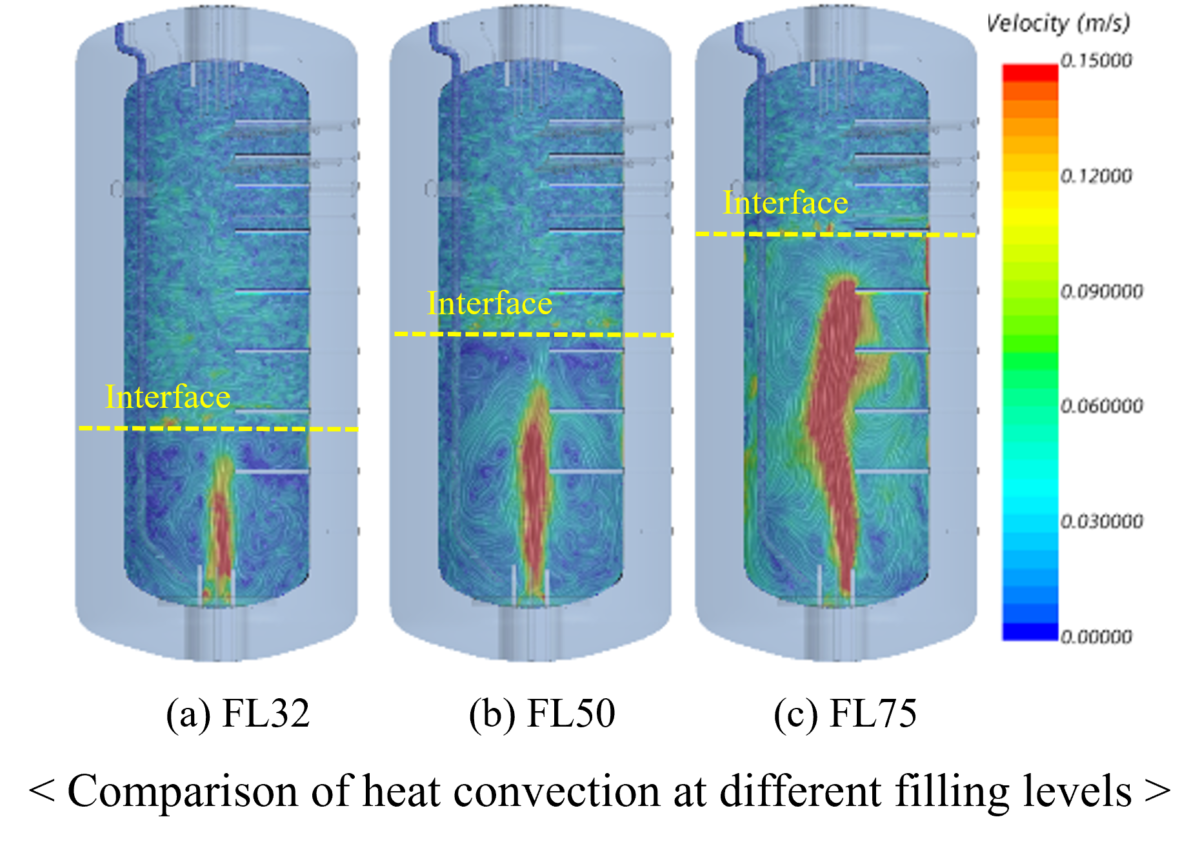
The South Korean research team, led by Jong-Chun Park, looked at how BOG varies with the tank filling ratio (FR), which is the ratio of the mass of liquefied fuel in the tank to the capacity of the tank at 15 C. They also used liquefied nitrogen instead of hydrogen in the experiment to validate simulation results and reduce safety risks.
“It was observed that the amount of BOG tends to increase in the form of a quadratic function based on the trend line, as FR increases,” they said, noting that vaporization increases drastically as the tanks become emptier.
Cryogenic fuel tanks maintain temperatures below -253 C to transport hydrogen as a liquid, overcoming the poor safety and storage efficiency of the current hydrogen storage methods based on high-pressure gas. The energy to cool hydrogen to a cryogenic liquid state equates to about 35% of hydrogen’s total energy.
Two primary alternative storage methods are liquid organic hydrogen carrier (LOCH) storage and conversion into ammonia. According to the researchers, the main advantage of these two methods is that the hydrogen can be stored at room temperature. The disadvantages are high energy requirements and difficulty using them for high-purity hydrogen.
The group explained that both techniques are in the early stages of development and are not ready for commercialization.
This content is protected by copyright and may not be reused. If you want to cooperate with us and would like to reuse some of our content, please contact: editors@pv-magazine.com.

By submitting this form you agree to pv magazine using your data for the purposes of publishing your comment.
Your personal data will only be disclosed or otherwise transmitted to third parties for the purposes of spam filtering or if this is necessary for technical maintenance of the website. Any other transfer to third parties will not take place unless this is justified on the basis of applicable data protection regulations or if pv magazine is legally obliged to do so.
You may revoke this consent at any time with effect for the future, in which case your personal data will be deleted immediately. Otherwise, your data will be deleted if pv magazine has processed your request or the purpose of data storage is fulfilled.
Further information on data privacy can be found in our Data Protection Policy.