Most perovskite materials that have been the subject of research for applications in solar are based on highly toxic lead, and also organic materials, which often cause issues with stability.
Now, researchers from Brown University and the University of Nebraska – Lincoln, have developed inorganic perovskite solar cells, which substitute lead for titanium. Research published in the journal Joule demonstrates that this material could be useful in tandem cell arrangements with silicon or other solar cell materials.
“Titanium is an abundant, robust and biocompatible element that, until now, has been largely overlooked in perovskite research,” said Professor in Brown’s School of Engineering Nitin Padture. “We showed that it’s possible to use titanium-based material to make thin-film perovskites and that the material has favorable properties for solar applications which can be tuned.”
The cells produced were semi transparent, with a bandgap of 1.8 eV, and efficiency of 3.3%. This is well below results achieved with lead based perovskites, and even further from commercial efficiency levels, but the researchers are keen to stress that this is a good result for the first time working with a new material, and that there is plenty of room for improvement.
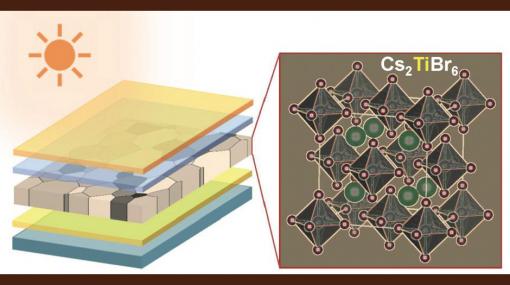
The research began with computer simulations carried out by theoreticians at the University of Nebraska – Lincoln, which showed the potential of a class of perovskites based on cesium, titanium and a halogen, such as bromine or iodine.
Other attempts to create lead free perovskites have primarily focused on tin, which the researchers say is a weaker candidate than titanium, as it is more prone to rust.
The material also exhibited open circuit voltage of more than 1 V, which the researchers said is further evidence of the material’s potential. “Open circuit voltage is a key property that we can use to evaluate the potential of a solar cell material,” said Padture. “Having such a high value at the outset is very promising.”
This content is protected by copyright and may not be reused. If you want to cooperate with us and would like to reuse some of our content, please contact: editors@pv-magazine.com.



A pity they chose to publish in Elsevier’s Joule, and not an open-access journal.