As our understanding of perovskite solar cells improves, scientists are looking deeper and deeper into the material to observe the structure, the way electrons flow through it, and any obstacles limiting the performance.
Defects in the material, particularly at the boundaries between crystal grains, have been shown to cause problems, causing the structure to quickly degrade when exposed to UV light or moisture. Among the many approaches suggested to solve this, doping the cells with chlorine has shown significant improvements.
However, scientists have struggled to explain why chlorine had these effects, or whether it even remained present in the material after deposition. This was the starting point for a group of scientists led by the Okinawa Institute of Science and Technology (OIST). “The research community had no idea why they were seeing these improvements,” explained OIST scientist Afshan Jamshaid. “Once added, the researchers couldn’t track the chlorine – they couldn’t tell whether the chlorine was incorporated deep into the perovskite material, stayed at the surface or even left the material during the manufacturing process.”
Confirming chlorine
Aiming to provide a definitive answer, Jamshaid and the group set out to fabricate chlorine doped perovskite solar films and pry into their structure right down to the atomic level, to confirm the presence or absence of chlorine. Using an imaging technique called scanning tunnelling microscopy, they observed dark depressions on the surface of the films, which were not present in samples without the chlorine doping.
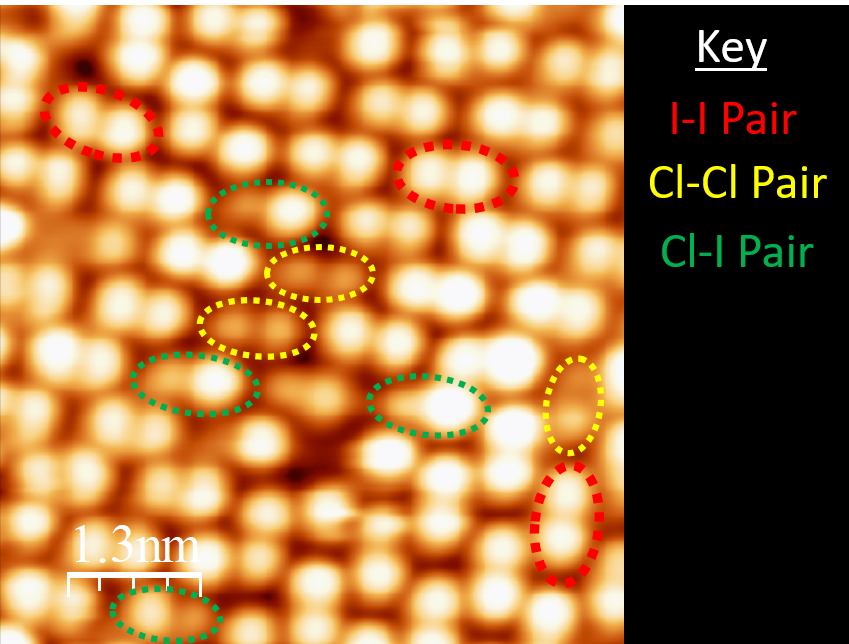
Image: OIST
They were then able to calculate (with the help of collaborators at China’s Soochow University) that these dark patches were spots where chlorine had replaced iodine in the perovskite structure. And since more chlorine appeared to be present at the grain boundaries, they now suspect that chlorine serves to reduce the number of surface defects in the material, increasing its stability. Their work is described in full in the paper Atomic-scale insight into the enhanced surface stability of methylammonium lead iodide perovskite by controlled deposition of lead chloride, published in Energy & Environmental Science.
Concentration
The group also trialled different deposition times, varying the final concentration of chlorine in the material. Through this, they are able to claim that 14.8% represents the ideal surface concentration of chlorine to boost stability. Any lower, and the improvements would be less pronounced, any higher and the chlorine will start to drastically alter other properties of the perovskite.
They plan to continue investigating how chlorine behaves as a dopant for perovskites, with the next step to create a fully operational solar cell based on a chlorine doped perovskite layer. “…fundamental studies like these are so important – they help device engineers pin down the most optimal manufacturing process without as much trial and error,” said Jamshaid. “By understanding how the dopants improve the material, it can also guide us towards new chemical mixes that might work even better.”
This content is protected by copyright and may not be reused. If you want to cooperate with us and would like to reuse some of our content, please contact: editors@pv-magazine.com.




By submitting this form you agree to pv magazine using your data for the purposes of publishing your comment.
Your personal data will only be disclosed or otherwise transmitted to third parties for the purposes of spam filtering or if this is necessary for technical maintenance of the website. Any other transfer to third parties will not take place unless this is justified on the basis of applicable data protection regulations or if pv magazine is legally obliged to do so.
You may revoke this consent at any time with effect for the future, in which case your personal data will be deleted immediately. Otherwise, your data will be deleted if pv magazine has processed your request or the purpose of data storage is fulfilled.
Further information on data privacy can be found in our Data Protection Policy.