Among the many different routes pursued by researchers to improve on today’s battery energy storage technologies, solid-state batteries could be the most promising. And many in the industry are optimistic that the approach will eventually bring about a “step change” in battery technology, greatly improving the capacity, lifetime and safety of lithium-ion batteries.
Swapping out the current generation of liquid/gel electrolytes with a solid material could eliminate any fire risk from the battery, as well as improving energy-to-weight ratios and removing the additional “energy redundant” packaging materials required to hold a liquid, among several other advantages.
The key development though is that the solid electrolyte would allow for the use of a lithium-metal anode, with far higher energy density than the graphite that’s commonly used today. Though there is research into integrating lithium-metal with liquid or semisolid electrolytes, all-solid-state is viewed by many as the best approach.
But lithium metal's tendency to form dendrites – branching structures that grow from the anode into the electrolyte and can cause short circuits and battery failure – is an issue for solid-state batteries as well. With ceramic-based solid electrolytes, dendrite formation can lead to cracks in the electrolyte and short-circuiting in the battery cell.
Observing batteries at work
Given the solid nature of the battery and the tiny scale at which these mechanisms occur, observing and understanding how they progress is a difficult task. A group of scientists led by the UK-based Faraday Institution seeks to address this in new research, where they use sophisticated imaging techniques to observe the batteries in operation, building on earlier insights gained from imaging the batteries after operation. “Ex-situ and destructive methods, involving sectioning the ceramic electrolyte and imaging the lithium metal within using techniques such as scanning electron microscopy, have proved valuable in demonstrating the problem,” the group explains. “However, it is important to follow the development of cracks and the ingress of lithium metal into the ceramic during operation of the cell in order to understand the mechanism of dendrite penetration and cell failure.”
Using X-ray computed tomography – an imaging technique similar to the CAT scans used in medicine – the group followed the progression of cracking and lithium ingress into the electrolyte, in a battery comprising a solid argyrodite (Li6PS5Cl) electrolyte sandwiched between two lithium metal electrodes. Their results are found in the paper Visualizing plating-induced cracking in lithium-anode solid-electrolyte cells, published in Nature Materials.
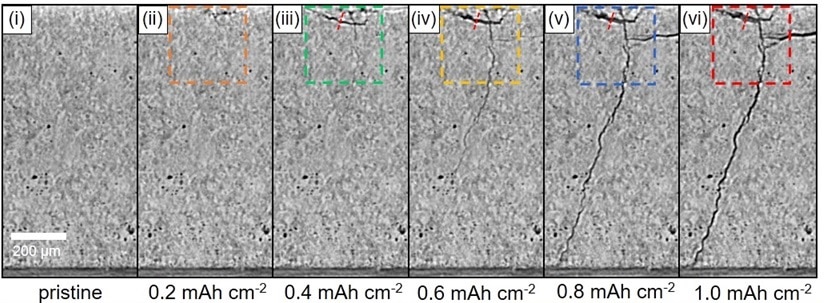
Image: Faraday Institution
Their observations confirmed that the cracks propagate through the electrolyte ahead of the lithium dendrite, proceeding along a path where the electrolyte porosity is above the material’s average. “Continued Li ingress widens the cracks and drives their propagation, but from the rear, not the crack tip,” the group explains. They note that even once a crack grows the whole way across the electrolyte, no short-circuiting occurs until later on when lithium has filled the crack completely.
“We show that cracks propagate through the electrolyte far ahead of the lithium dendrites, rather than the lithium metal driving the crack tip forward,” the group explains. By confirming that dendrites drive the crack formation from the base of the crack and that no lithium metal is present in the tip of the crack, the group is able to suggest better strategies to suppress both mechanisms. “Our findings suggest focussing more attention on blocking dry crack propagation to block dendrite propagation, for example by ceramic toughening and crack block-ing, including strategies such as fiber reinforcement and transformation toughening”, they conclude.
This content is protected by copyright and may not be reused. If you want to cooperate with us and would like to reuse some of our content, please contact: editors@pv-magazine.com.




100 comments
By submitting this form you agree to pv magazine using your data for the purposes of publishing your comment.
Your personal data will only be disclosed or otherwise transmitted to third parties for the purposes of spam filtering or if this is necessary for technical maintenance of the website. Any other transfer to third parties will not take place unless this is justified on the basis of applicable data protection regulations or if pv magazine is legally obliged to do so.
You may revoke this consent at any time with effect for the future, in which case your personal data will be deleted immediately. Otherwise, your data will be deleted if pv magazine has processed your request or the purpose of data storage is fulfilled.
Further information on data privacy can be found in our Data Protection Policy.