A team of scientists working at Oxford University in the U.K. have made valuable insights into the performance of solid-state lithium-ion batteries.
By improving our understanding of the mechanisms that cause solid state batteries to fail under frequent cycling (a barrier to commercial production), the research could allow scientists to pursue new battery designs that avoid or mitigate these failure mechanisms.
In a paper, “Critical stripping current leads to dendrite formation on plating in lithium anode solid electrolyte cells,” published in the journal Nature Materials, the scientists observed the formation of dendrites – branching networks of lithium that grow through the electrolyte – and voids, which reduce the area of contact between the anode and the electrolyte.
“It is key to break down the scientific barriers that prevent the progression to market of technologies that will enable our vision of the future of mobility. The study by Oxford researchers is one early example of a scientific advance that the Faraday Institution was set up to drive,” said Tony Harper, director of the ISCF Faraday Battery Challenge at U.K. Research & Innovation.
The researchers worked with a three-electrode battery, in order to better be able to separate lithium plating from stripping, using Argyrodyte (Li6PS5Cl) as an electrolyte.
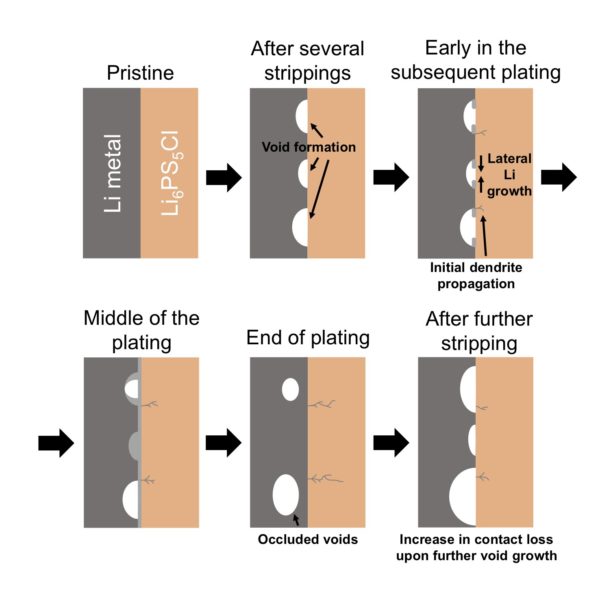
They found that keeping current density low is key to avoiding these undesirable mechanisms. “When the stripping current density removes Li from the interface faster than it can be replenished, voids form in the Li at the interface and accumulate on cycling, increasing the local current density at the interface and ultimately leading to dendrite formation on plating, short circuit and cell death,” explains the paper’s abstract. “This occurs even when the overall current density is considerably below the threshold for dendrite formation on plating.”
This content is protected by copyright and may not be reused. If you want to cooperate with us and would like to reuse some of our content, please contact: editors@pv-magazine.com.




2 comments
By submitting this form you agree to pv magazine using your data for the purposes of publishing your comment.
Your personal data will only be disclosed or otherwise transmitted to third parties for the purposes of spam filtering or if this is necessary for technical maintenance of the website. Any other transfer to third parties will not take place unless this is justified on the basis of applicable data protection regulations or if pv magazine is legally obliged to do so.
You may revoke this consent at any time with effect for the future, in which case your personal data will be deleted immediately. Otherwise, your data will be deleted if pv magazine has processed your request or the purpose of data storage is fulfilled.
Further information on data privacy can be found in our Data Protection Policy.